Ever wondered if one day you could design a drug the way you build a startup, fast, smart, and data-driven? AI drug discovery is turning that idea into reality. Well, that future isn’t knocking on the door anymore; it’s already inside the room.
With AI-powered Software-as-a-Service (SaaS) platforms, scientists can now design molecules, analyze massive biological datasets, identify precise drug targets, and even simulate outcomes before stepping into a lab. This shift is redefining how medicines are created, where speed, precision, and data-driven decisions are replacing traditional trial-and-error approaches. What once sounded like science fiction is quickly becoming standard practice in the pharmaceutical industry.

For decades, the drug development process has been one of the most expensive and time-consuming journeys in modern science. Pharmaceutical companies invested billions of dollars and spent over 10–15 years developing a single drug, yet most candidates never made it to market. The process was slow, uncertain, and heavily dependent on manual experimentation.
But now, the game has changed. Artificial intelligence, combined with SaaS healthcare platforms, is creating a smarter and more efficient ecosystem. Leading players like Insilico Medicine, Recursion Pharmaceuticals, and Exscientia are proving that AI in pharma is not just a trend. It is a structural shift shaping the future of medicine.
Also Read:
- Gene Therapy Commercialization: The Role of Strategic Storytelling
- Women Riders in India Changing the Biking Landscape
Traditional Drug Discovery: A Long, Costly Road in the Drug Development Process
The drug development process has long been slow, expensive, and uncertain, relying on sequential stages and trial-and-error methods that demand years of research, high investment, and still face high failure rates.

Disease Identification to Hit Discovery
This is where everything begins. The process begins with disease identification. Researchers start by understanding what is going wrong inside the human body. From there, the focus shifts to target identification, pinpointing a specific protein, gene, or biological pathway responsible for the disease.
Once a target is identified, the next phase, hit discovery, feels a bit like searching for a needle in a haystack. Thousands, sometimes millions, of chemical compounds are tested just to find a few that show even the slightest interaction with the target. Most of them go nowhere.
Lead Optimization to Clinical Trials
Now comes refinement. Those few “hits” then move into lead optimization, where chemists refine them, tweaking their structure to improve effectiveness and reduce toxicity. This stage demands patience, repeated lab work, and, quite frankly, deep pockets.
If a compound shows promise, it enters preclinical testing, where it is tested in lab environments and animal models to evaluate safety. Only after clearing these stages, it moves into clinical trials, where it is tested on humans in multiple phases. Phase I focuses on safety, Phase II on effectiveness, and Phase III on large-scale validation. Even here, the odds are brutal, with most drugs failing after years of investment.
The traditional model has delivered results, but its limitations in speed, cost, and efficiency are driving the shift toward AI in pharma and more advanced approaches.
Clinical Trials to Market Launch in the Drug Development Process
The final stages of the drug development process involve strict regulatory review and controlled market entry, where safety, efficacy, and compliance determine whether a drug can be approved and made available to patients.
Clinical Trial and Regulatory Approval
If a drug survives this gauntlet, it moves to regulatory approval, where authorities carefully review every piece of data. Extensive documentation, attention to detail, and safety data must be reviewed and approved by regulatory authorities.
Regulatory Approval to Market Launch
Only after passing this final checkpoint does it reach market launch, where it is manufactured, distributed, and finally prescribed.
Timeline and Uncertainty in the Drug Development Process
From start to finish, this journey can take over a decade. And even then, success is never guaranteed. It’s no surprise that the industry has long been searching for a better way.
Traditional Drug Discovery Stages Overview
This overview outlines the structured progression of the drug development process, from disease identification to market launch, where each stage builds on the previous one, making it a linear, time-intensive system with high costs, risks, and significant uncertainty.

Traditional Drug Designing Flow Chart
This flow chart represents the step-by-step progression of the drug development process, from disease identification to market launch, highlighting its linear, time-intensive nature and dependency on sequential validation at each stage.
Drug discovery has always been a process of elimination, not creation.
What Does the Traditional Drug Designing Flow Chart Represent?
The Traditional Drug Designing Flow Chart represents a step-by-step, linear model of the drug development process, where each stage depends on the previous one, highlighting time intensity, cost, and sequential validation.
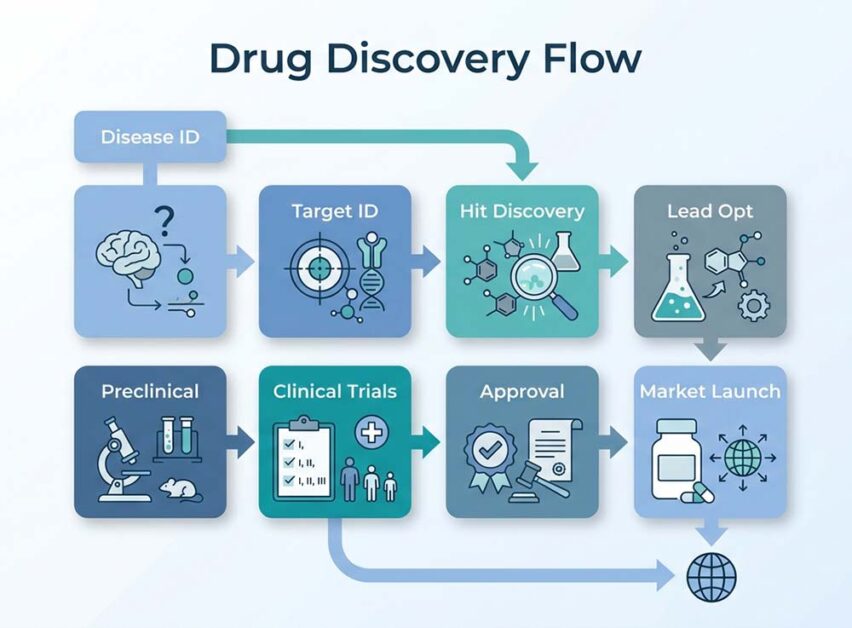
These final stages highlight why the traditional drug development process is often slow, expensive, and uncertain, which is driving the growing shift toward AI in pharma and SaaS healthcare platforms for faster innovation.
AI Drug Discovery vs Traditional Drug Development Process in Pharma
The comparision highlights how AI drug discovery is transforming the traditional drug development process by improving speed, reducing costs, and enabling scalable innovation through SaaS healthcare platforms, making drug creation more efficient and globally accessible.
| Factor | Traditional Drug Discovery | AI-Powered SaaS Drug Discovery |
| Time to Market | 10-15 years | 3-6 years |
| R&D Cost | $1-12 billion | Reduced Significantly |
| Efficiency | Low | High |
| Scalability | Limited | Cloud-Based, global |
The shift from traditional systems to AI in pharma is not theoretical. It is already visible through companies adopting data-driven models, automation, and SaaS healthcare platforms to accelerate drug pipelines and improve commercial outcomes.
Real-World Companies and Revenue Models
| Company | Revenue | AI-Stage usage | Drugs | Impact |
| Insilico Medicine | ~$50 M | End-to-end AI pipeline | INS018_055 | Multi-billion-dollar collaborations, SaaS licensing |
| Recursion Pharmaceuticals | ~$200M | Preclinical + trials | Multiple pipelines | Closed-loop robotic labs, AI-driven trials |
| Exscientia | ~$120M | Drug design | DSP-1181 | Fastest AI drug design, licensing & partnerships |
| BenevolentAI | ~$300M est. | Target discovery | COVID repurposing | Knowledge graph AI, SaaS platform access |
These comparisons clearly show how AI in pharma is reshaping efficiency, cost structures, and scalability across the drug development ecosystem.
Challenges and Future of AI Drug Discovery in Pharma
AI drug discovery is advancing rapidly, but it still faces critical challenges around data reliability, regulation, and biological complexity, even as SaaS healthcare platforms and AI in pharma continue to push the boundaries of speed, precision, and innovation.
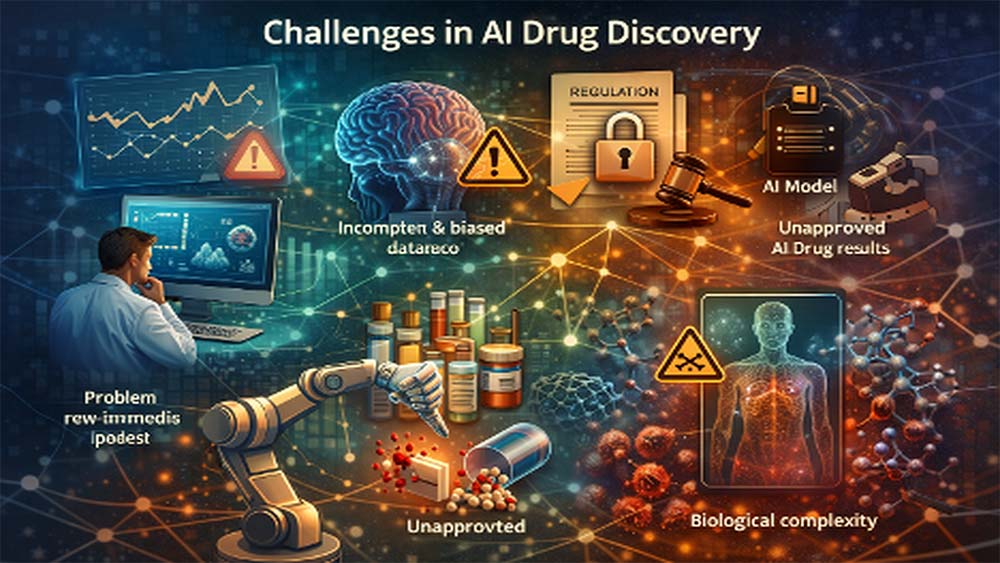
Key Challenges in AI Drug Discovery
Of course, every revolution comes with its own set of challenges, and AI is no exception. One of the biggest hurdles is data quality. AI systems are only as reliable as the data they are trained on. Incomplete or biased datasets can lead to flawed predictions, which is a serious concern in healthcare.
Then there’s the issue of regulation. AI models often function as “black boxes,” making it difficult to explain how decisions are made. Regulatory bodies need transparency, and without it, approvals can slow down. Another reality check is that very few AI-designed drugs have been fully approved so far, showing that the field is still maturing.
Biological complexity remains a major limitation. The human body does not always follow predictable patterns, and some AI-designed drug candidates fail in later stages due to unexpected toxicity. This reinforces that AI in pharma currently works best alongside human expertise, not as a replacement.
Why Does AI Drug Discovery Still Face Approval and Accuracy Challenges?
AI drug discovery faces approval and accuracy challenges due to limited high-quality datasets, lack of model transparency, evolving regulatory frameworks, and the unpredictable complexity of human biology, which makes consistent validation and large-scale clinical success difficult.
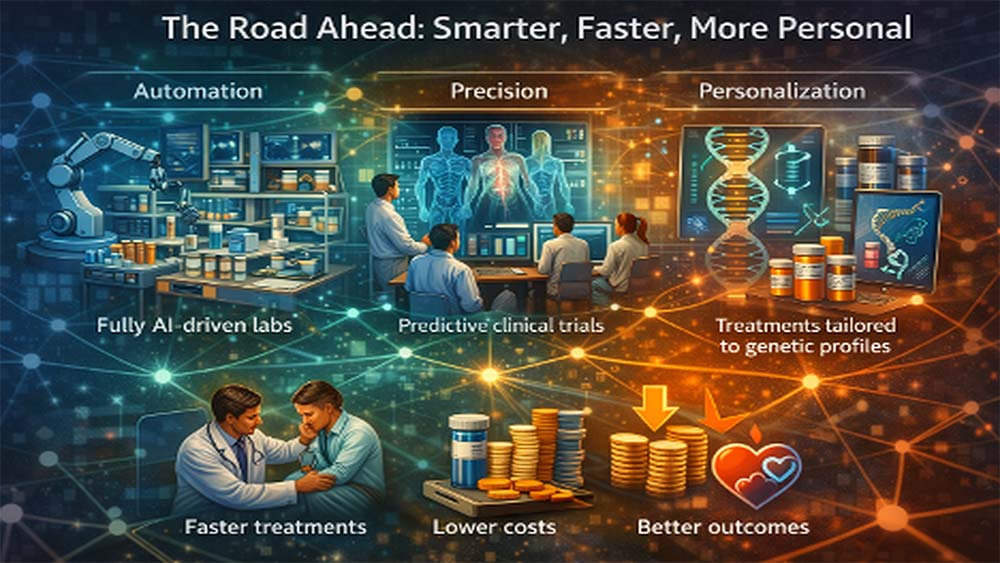
Despite these challenges, the future is moving toward smarter, faster, and more personalized drug development, where AI drug discovery continues to improve outcomes while working alongside scientific expertise.
Also Read:
Conclusion
AI-powered SaaS is not just improving AI drug discovery; it is redefining the entire drug development process. What was once slow, expensive, and uncertain is becoming faster, smarter, and more precise through AI in pharma and SaaS healthcare platforms. Companies like Insilico Medicine, Recursion Pharmaceuticals, and Exscientia are already proving how data-driven innovation can accelerate outcomes.
The real shift lies in collaboration. AI is not replacing scientists but enhancing their decision-making and reducing risk. As this ecosystem evolves, the focus remains clear: better treatments, faster access, and more reliable outcomes that ultimately improve patient lives.
Because in the end, the goal isn’t just better technology.
It’s better outcomes, better treatments, and ultimately, better lives.
✍️ Guest Write with Us
At TrendVisionz, the digital venture of Nuteq Entertainment Pvt Ltd, we welcome contributors who share our passion for storytelling, digital marketing, and innovation. Whether it’s an article, case study, or industry insight that educates, inspires, or entertains — we’d love to feature your perspective.
- Explore our work: Nuteq Entertainment | TrendVisionz | Author Profile
- Read our newsletters: Transforming Lives | BizTech Chronicle | Nuteq Newsline | Growth Catalyst
- Guest Contributions: BizCatalyst360 | Praja Today
- Books: Go Mindfulness: Practices for Professionals Coached
- Join the community: LinkedIn Group – Digital Marketing & Content Creation World
- Connect with me: LinkedIn | Twitter | #StoryforBusiness
- Share your review: Leave your review on Google — your feedback helps us grow.
Follow me on Twitter or LinkedIn. Check out our website.
How to Contribute:
Send your pitch or draft to Editor: anujmahajan@trendvisionz.com
References:
- Paul, S. M., Mytelka, D. S., Dunwiddie, C. T., et al. (2010). How to improve R&D productivity: the pharmaceutical industry’s grand challenge. Nature Reviews Drug Discovery. https://pubmed.ncbi.nlm.nih.gov/21091654/
- Hughes, J. P., Rees, S., Kalindjian, S. B., & Philpott, K. L. (2011). Principles of early drug discovery. British Journal of Pharmacology. https://pmc.ncbi.nlm.nih.gov/articles/PMC3058157/
- Mak, K. K., & Pichika, M. R. (2019). Artificial intelligence in drug development: present status and future prospects. Drug Discovery Today.
- Vamathevan J, Clark D, Czodrowski P, Dunham I, Ferran E, Lee G, Li B, Madabhushi A, Shah P, Spitzer M, Zhao S. Applications of machine learning in drug discovery and development. Nat Rev Drug Discov. 2019 Jun;18(6):463-477. doi: 10.1038/s41573-019-0024-5. PMID: 30976107; PMCID: PMC6552674.
- National Center for Biotechnology Information (NCBI). Drug Discovery and Development Process.
- Frontiers in Drug Discovery. (2023). Introduction to small molecule drug discovery and preclinical development.
- Insilico Medicine Official Website- https://insilico.com/
- Recursion Pharmaceuticals Official Website: https://www.recursion.com/
- DeepMind – AlphaFold Research: https://www.deepmind.com/research/highlighted-research/alphafold


